Mirrors of Medicine TM
A unique learning method to apply new insights to daily clinical practice
The overwhelming amount of new insights in medicine and healthcare calls for time-efficient and learner-centric ways of CME for addressing the need of continuing professional development. The Mirrors of Medicine™ method uses innovative methods for both developing learning materials and measuring their impact:
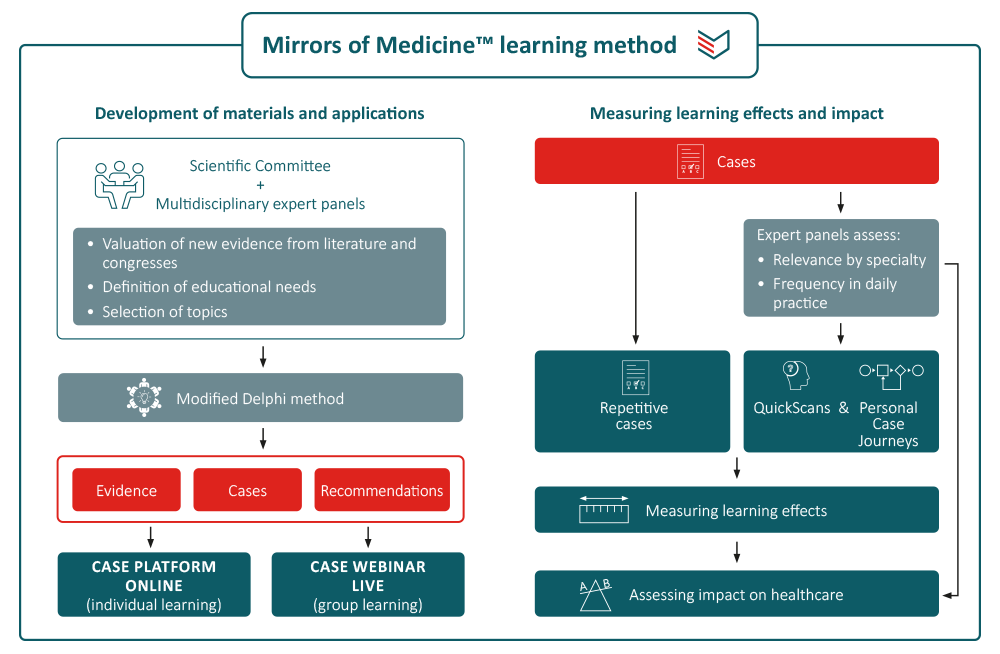
Development of cases and related materials
Topics are selected and defined by the Scientific Committee based on educational needs in a specific disease area. Cases and feedback are prepared and reviewed by physicians involved in clinical decision making in the topic under consideration. Clinical options within cases are validated by a multidisciplinary expert panel (9–15 members), using the RAND/UCLA Appropriateness Method [1,2]. The appropriateness of a clinical option is expressed on a 9-point scale ranging from inappropriate (1–3) via equivocal/uncertain (4–6) to appropriate (7–9). All materials are periodically reviewed and updated when new insights become available. Where needed, re-ratings are performed. All materials are reviewed and approved by the expert panel.
Learning methods and impact measurement
Users have different options to learn what they want and need to learn. They can subscribe to the weekly case series to receive cases by email and get immediate feedback. They can also follow a CME course consisting of a series of cases and an assessment. Successful completion earns 1 European credit point. The most innovative approach is the QuickScan that identifies individual learning needs in a specific disease area, followed by automated personalised case journeys to address the learning gaps. A recent randomised study showed that both non-individualised and personalised learning on the PinPoint Case Platform are effective methods, but that the personalised approach required significant less time. [3] An extensive programme amongst urological residents showed an impressive short-term learning effect (from 48% to 93% correct answers) with a drop to 67% at 3 months follow-up. Nevertheless, there remained a net increase of correct answers of around 40%. [4]
- Brook RH, Chassin MR, Fink A, et al. A method for the detailed assessment of the appropriateness of medical technologies. Int J Technol Assess Health Care 1986;2:53-63.
- Fitch K, Bernstein SJ, Aguilar MD, et al. The RAND/UCLA Appropriateness Method. Santa Monica: RAND, 2001.
- Stoevelaar H, Bahl A, Helsen N, et al. Personalised versus non-individualised case-based CME: A randomised pilot study. J Eur CME 2022;11(1):2153438.
- Stoevelaar H, Van Ruysevelt L, Yperman J. PinPoint Case Platform. Annual Report 2022. ISSECAM, Lier (Belgium), December 2022.
